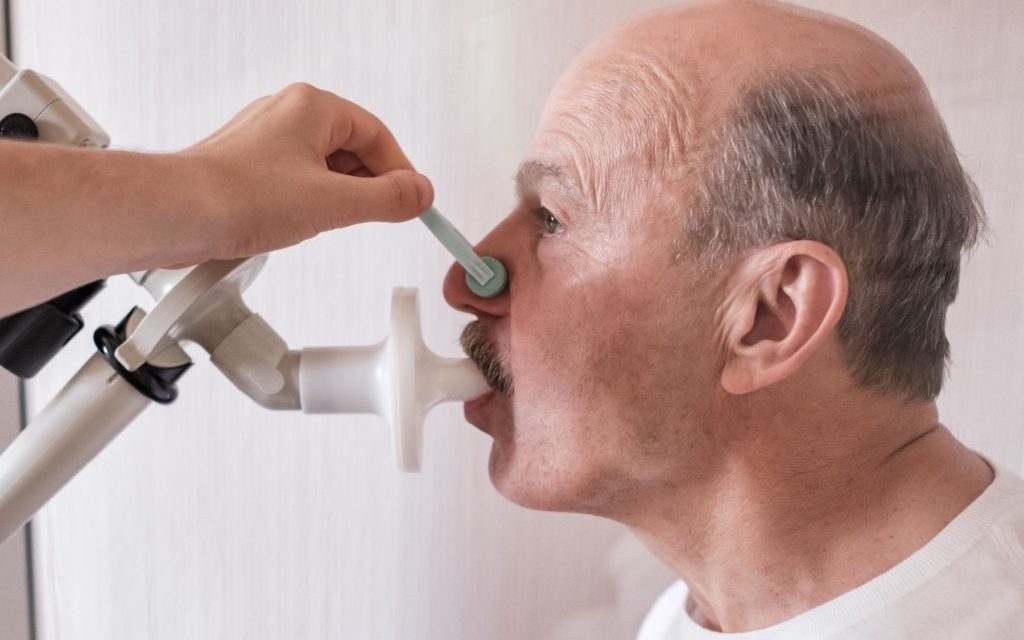
You can have a role in research that could change medicine forever.
Paid clinical research studies are being conducted at Heart Lung Centre to learn more about investigational medications that are not yet available to the public.
Are Clinical Trials safe?
This is a very important question. At Heart Lung Centre, all our trials are reviewed by an independent Research Ethics Committee, who ensure that our trials are as safe as possible. We are here to answer any questions.
Learn More

Do I get paid?
Our trials vary greatly in terms of time spent on the unit and the number of procedures performed. Once we have identified a suitable trial, we can inform you of the potential compensation for your participation.
Learn More

What facilities are there?
Whether you’re here for an hour or the whole day, we will endeavour to make your stay as comfortable as possible. The volunteer lounge will provide you with a space to pass the time pleasantly between assessments.
Why do you offer Clinical Trials?
Here at Heart Lung Centre we adhere to MHRA (The Medicines and Healthcare products Regulatory Agency) who are responsible for regulating the UK medical devices market. You can find out more information here.
We do not have any studies for healthy volunteers.
-
Asthma studies
Asthma Clinical Trials are to evaluate newly developed medications and new combinations of well-established medications for the treatment of asthma.
To take part in our studies you:
- Must have been diagnosed with Asthma for 12 months or more
- Must not be, pregnant, lactating or trying to conceive with your partner (if applicable)
- Must not be currently enrolled in another clinical trial
For all studies, participants are reimbursed for their time, travel and inconvenience. If you would like to find out more register your interest and we will be in touch. Or contact us on 0207 034 3341 or recruitment@heartlungcentre.com.
Even if you do not believe you may be eligible, do register your interest as new opportunities are pending.
Current studies are detailed below.
-
AsthmaStudy: Sanofi
This is a Phase II, randomised, double-blinded, placebo-controlled clinical trial to assess the safety and efficacy of a new study drug. This drug is designed to inhibit the initiation and amplification of the asthmatic response. It has been assessed in two phase 1 studies. It was found to be well tolerated by both healthy participants and asthmatics.
In total, there will be 18 visits across 14 months.
Pre-screening: We will ask you some questions about your medical history and conduct one lung function assessment. (If you screen fail- £50 participant reimbursement)
Screening: If you pass the Pre-screening, we will complete the screening visit the same day. This includes ECG, Vital signs, blood tests and further lung function tests. (£70 participant reimbursement)
Run in Visit: You will be issued an e-diary, home spirometer and a new inhaler to replace your current asthma medication. The new inhaler is an approved asthma maintenance inhaler, but it will be a higher dose than your current one.
Randomisation: Assessments include: bloods, ECG vital signs and lung function. If the new inhaler has not significantly improved the results of one of your lung function assessments, you will be considered a candidate for the study drug and will be randomized into the trial.
Once randomised you will be brought back to the clinic every month to receive the study drug until the end of the study. The study drug will be administered via subcutaneous injection. During this time, you will remain taking the new inhaler, using the study diary and at home spirometer.
You will be reimbursed for your Travel up to £100 for each visit (upon evidence of receipts).
For your time and participation, you will also be reimbursed £70 per visit you attend (£1,260 total). NB. Participants must agree to use contraception, and refrain from donating sperm or eggs throughout the trial and for an additional 5 months after the last dose.
To take part in this study you:
- Must have been diagnosed with Asthma for 12 months or more
- Must be between the ages of 18 to 80
- Must use preventative Asthma medication to manage your Asthma for at least 1 month
- Must have had an asthma exacerbation in the last year that required oralsteroids for at least 3 days
- Must be a non-smoker or an ex-smoker for at least 6 months
If you believe you may be eligible to take part and would like to find out more, register your interest on /clinical-trials-request/ and we will be in touch. Or contact us to find out more on 0207 034 3341 or at recruitment@heartlungcentre.com.
Even if you do not believe you may be eligible, do register your interest as new opportunities are pending.
REGISTER

-
COPD studies
Chronic Obstructive Pulmonary Disease Clinical Trials are to evaluate newly developed medications and new combinations of well-established medications for the treatment of COPD.
To take part in our studies you:
- Must not be, pregnant, lactating or trying to conceive with your partner (if applicable)
- Must not be currently enrolled in another clinical trial
For all studies, participants are reimbursed for their time, travel and inconvenience. If you would like to find out more register your interest and we will be in touch. Or contact us on 0207 034 3341 or recruitment@heartlungcentre.com.
Even if you do not believe you may be eligible, do register your interest as new opportunities are pending.
Current studies are detailed below.
-
COPD Study: Uniquity/DevPro
This is a Phase 2, randomized, double-blind, placebo-controlled clinical study to evaluate the safety, tolerability, immunogenicity of the study drug versus a placebo on top of standard of care in participants with COPD.
This medication has previously been assessed in two clinical studies, one in healthy participants and one in participants with atopic dermatitis. Based on previous research studies, the risks associated with study treatment are expected to be limited to local injection site discomfort in the majority of cases.
Your participation in this study will last about 32 weeks and include approximately 11 visits to the study site. Each visit will last between 3-5 hours. To assess your eligibility, the screening visit will include questions about your medical history, a physical assessment, vital signs, ECGs, blood tests, urine sampling and Lung Function testing.
You may also be required to undergo a CT scan of the chest/lungs if you have not had one within the last 3 months.
If you are found to be eligible, you will then go into a 12-week treatment period followed by a 16- week follow-up period. In the treatment phase of the study, you will be administered 2 subcutaneous injections on Day 1, Week 4, and Week 8 and be required to stay in the hospital for 2-4 hours post-drug administration.
For your time and inconvenience, you will be reimbursed £150 per visit you attend, totalling up to £1,500. Your travel will also be reimbursed up to £200 per visit.
To take part in this study you:
- Must have been diagnosed with COPD for 12 months or more.
- Must be aged between 40 and 75.
- Must use a daily preventative inhaler (non-blue inhaler) for at least 3 months.
- Must be an ex-smoker for a minimum of 6 months.
If you believe you may be eligible to take part and would like to find out more, register your interest on /clinical-trials-request/ and we will be in touch. Or contact us to find out more on 0207 034 3341 or at recruitment@heartlungcentre.com .
Even if you do not believe you may be eligible, do register your interest as new opportunities are pending.
REGISTER -
COPD Study: Oratos
This study is a randomised, double-blinded, placebo-controlled crossover study to assess the effect of Trixeo inhaler versus placebo on cardiac and lung function in participants with COPD and hyperinflation.
This triple-acting inhaler contains Budesonide/Glycopyrronium/Formoterol Fumarate and is currently approved for the treatment of COPD in several countries. 5 phase III studies have indicated that this inhaler is safe and well tolerated.
In total, there will be 6 visits over 11 weeks, with visits every 2-3 weeks. You will receive both the placebo inhaler and study drug in separate 3-week treatment periods, where you will take the medication 2 puffs twice a day, in the morning and evening. Additionally, all maintenance inhalers would need to be stopped throughout the study duration, but you will be free to use Salbutamol as needed. If you are on a prescribed LAMA inhaler, an additional SAMA, ipratropium bromide will be dispensed for use as needed.
At the screening visit, we will assess your eligibility by asking questions about your medical history, a physical assessment, vital signs, ECG, blood tests and lung function testing. If you are found to be suitable there will be a second screening visit, where you will undergo further lung function tests and vital signs, and will be issued and taught how to use your e-dairy.
During the trial, you will undergo three cardiac MRIs and three Echocardiograms with Medneo, our medical imaging partner based in Central London. Questions/ concerns can be discussed in person with our medical team. An MRI is a medical imaging technique that uses magnetic fields and radio waves to create detailed images of the inside of your body. Whilst an Echocardiogram is an ultrasound scan to look at your heart and nearby blood vessels.
For your time and inconvenience, you will be reimbursed £80 on your first visit. If you are found to be suitable and are enrolled into the study, you will be reimbursed up to £880. Your travel will also be reimbursed up to £100.
To take part in this study you:
- Must have a diagnosis of COPD.
- Must be aged between 40 and 80.
- Must use a daily preventative inhaler (non-blue inhaler) for at least 6 weeks.
- Must be a current or ex-smoker.
If you believe you may be eligible to take part and would like to find out more, register your interest on /clinical-trials-request/ and we will be in touch. Or contact us to find out more on 0207 034 3341 or at recruitment@heartlungcentre.com .
Even if you do not believe you may be eligible, do register your interest as new opportunities are pending.
REGISTER

-
Other Studies
In addition to our asthma and COPD studies, we are also conducting research into potential new therapies for other conditions.
If you would like to find out more register your interest and we will be in touch. Or contact us on 0207 034 3341 or recruitment@heartlungcentre.com.
Even if you do not believe you may be eligible, do register your interest as new opportunities are pending.
Current studies are detailed below.
-
Erectile dysfunction study: RestoreX
This research study is designed to evaluate the effectiveness of the RestoreX penile traction therapy (PTT) device in men who have undergone prostate surgery. Patients will be randomly divided into two groups: one group will use the RestoreX device daily for a specified period, while the other group will not use the device initially.
Throughout the study, patients will be asked to complete questionnaires assessing their sexual function and overall experience. The study will also track any changes in your health.
Participants who are initially assigned to the non-use group will have the option to start using the device at a later point in the study.
If you consent to take part in the study, during this visit we will record your concurrent conditions, and perform a physical (including an examination of the penis). You will also complete the first questionnaire. Please bring all medications you are currently using.
If you believe you may be eligible to take part and would like to find out more, contact us to find out more on 0207 034 3341 or email us at recruitment@heartlungcentre.com .
-
GSK - Cough Study
This is an observational study to measure cough variability in patients with Refractory or Unexplained Chronic Cough and those without a chronic cough.
This study will be conducted over a 4-week period (29 days). It involves an initial screening visit at our clinic, where you will be asked questions about your medical history.
If you are eligible, you will receive a cough-monitoring watch, which you will wear for 29 days, and be asked to download the Companion App on your smartphone. During this visit, you will receive onboarding information and training on how to use these.
The microphone in the watch continuously detects peak noises and determines whether it is a cough or non-cough. The watch sends only the data and time of the detected cough to the Companion app via Bluetooth. No sound or speech data is transmitted from the watch. You will also be asked to download another app to complete daily, weekly and fortnightly questionnaires on. At the end of the study period, participants will return the cough monitoring system to the site.
For your time and participation, you will be reimbursed £40 on your first visit. If you are found to be suitable and are enrolled in the study, you will be reimbursed for up to £860. Reasonable travel expenses may also be reimbursed.
Optional: There is also an optional concierge service which involves someone reaching out from the ICON CS team via telephone and/or email to assist you with certain study procedures or to follow up with you if there is any missing information for the study.
Optional- Chronic cough cohort only: At the first visit you will have the option to provide a saliva sample for future genetic research related to chronic coughs.
Note: You do not have to consent to either of these, to be part of the study.
If you’re interested and would like to find out if you’re eligible, please register your interest at/clinical-trials-request/ and we will be in touch. Or contact us to find out more on 0207 034 3341 or email us at recruitment@heartlungcentre.com.


FAQ'S
If you want to know more about clinical trials or are considering participating in one, the following frequently asked questions describe the role of a patient and will help you understand this unique research program.
FAQ'SLocation
Explore beyond the Heart Lung Centre and discover Respiratory Clinical Trials and our hospital Queen Anne Street Medical Centre. Our state of the art facility offers a range of medical services, including consultants in different specialties, diagnostic tests and an operating theatre.
13-14 Fitzroy Square,
London, W1T 6AH
E: recruitment@heartlungcentre.com
P: 0207 0343341
Give us a call or email us, we endeavour to answer all enquiries within 24 hours on business days.
We are open from 8am — 5pm week days.







